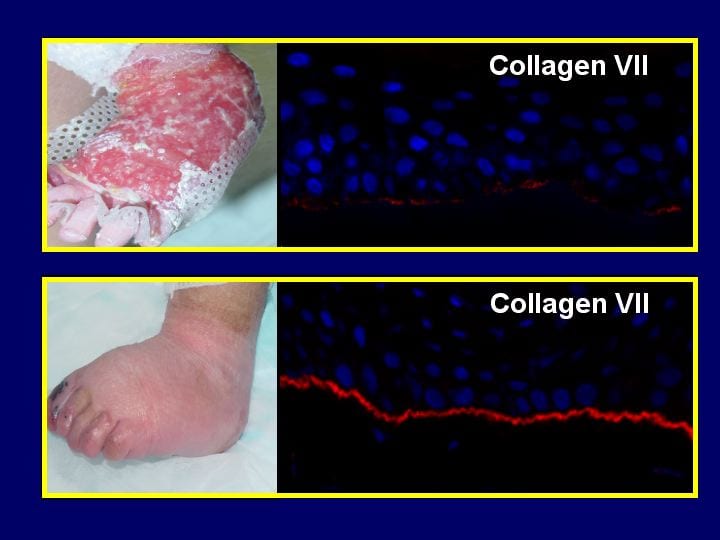
EB Research is carried out by dermatologists and scientists who are pioneering techniques with EB that are applicable to many other conditions.
There is a huge unmet clinical need for effective treatments for this devastating condition and research is now on the brink of delivering some.
Incurable genetic diseases
There are about 6000 human genetic diseases and in principle, the mutation cause of the majority of them could be correctable by genome editing and other forms of gene therapy. This is approximately 10% of people, 30 million in Europe alone. With advancing technologies, the new medical miracle will be treatments for these ‘incurable’ conditions.